
| Home |
| About Us |
| Patient Services |
| Physicians |
| XRay & MRI Review |
| News |
| Education |
| Links |
| Directions |
| Contact Us |
Hip Resurfacing |
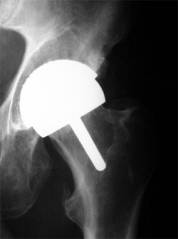
What Is It? Hip resurfacing arthroplasty has been present for many years outside of the United States but has seen limited use in the United States because of a historically high failure rate. In May 2006, the first hip resurfacing arthroplasty device received conditional FDA approval. This device, the Birmingham Hip Resurfacing arthroplasty, was permitted for use by U.S. surgeons who had received specialized training in either Canada or the United Kingdom. More recently, video conferencing training has been approved. Hip resurfacing arthroplasty is a form of joint replacement. The goals of hip resurfacing arthroplasty are to conserve the amount of bone removed during the surgery and limit the chances of a post-operative dislocation. A recent interest in hip resurfacing has emerged secondary to the historically high failure rates of traditional hip replacements in younger patients. Currently in the United States, hip resurfacing arthroplasty is considered a transitional device until a traditional total hip arthroplasty is needed. The true longevity of a hip resurfacing arthroplasty is still unknown, but limited studies have shown greater than 90% success at 5 years (i.e., less than 10% conversion to a traditional total hip arthroplasty). The basics of a hip resurfacing arthroplasty are similar to total hip replacement. One primary difference is that the surgical incision and exposure for a resurfacing procedure is twice as large because less of the femoral (“ball”) side of the joint is removed. The cup or socket side of a resurfacing is similar to a traditional total hip replacement. The acetabular cup is generally noncemented (“press-fit”) and the femoral component is generally cemented. Both the ball and socket are made out of a metal alloy (i.e., “metal on metal”). Special tools have been designed to improve access to the hip joint and facilitate component placement. The hip resurfacing procedure is more technically demanding than a traditional total hip replacement. Who Should Have It? Hip resurfacing arthroplasty is designed for younger patients whose activities may exceed the capabilities of a traditional total hip replacement (i.e., increased wear, dislocation risk, loosening). “Younger” is generally defined as less than 55 years old. Resurfacing technology can be used for a variety of hip conditions including osteoarthritis, traumatic arthritis, dysplasia, and avascular necrosis. The appropriateness of a resurfacing procedure may depend on the x-ray film appearance of the hip joint and the bone density of the patient. Women of child bearing age may not be candidates. What Are Some of The Risks? Basic surgical risks include infection, bleeding, and anesthesia. Other risks include damage to normal nerves, blood vessels, and surrounding muscle tissue. As the hip needs to be dislocated for the resurfacing, stretching may injure the sciatic nerve. The sciatic nerve controls the ability to move the foot. Other possible problems include blood clots, calcification of normal tissues (i.e., heterotopic calcification), intraoperative fracture, and dead hip bone. The primary mode of failure of hip resurfacing is fracture at the femoral neck. Failure of a resurfacing procedure necessitates placement of a traditional total hip replacement. An unknown risk of resurfacing is the long term release of metal ions from prosthetic wear, namely cobalt and chromium, into the body. This is not an all inclusive list.
|
Hip Resurfacing |
||
|
||
©2008-2014 wfuhip.com All Rights Reserved
The information on this Website is for general informational purposes only and SHOULD NOT be relied upon as a substitute for sound professional medical advice, evaluation or care from your physician or other qualified healthcare provider. If you have a medical problem or a health-related question, consult your physician or call Health On-Call at 336-716-2255 or 1-800-446-2255.